Description
Cardio Care Patient Monitor
Overview – Cardio Care Patient Monitor K12 & K15
The Cardio Care Patient Monitor K12 and K15are advanced multi-parameter bedside monitoring systems designed to provide continuous and accurate monitoring of vital signs in hospitals, intensive care units (ICUs), coronary care units (CCUs), operating theaters, emergency departments, recovery rooms, and general patient wards. These monitors combine advanced monitoring technology, user-friendly operation, and reliable performance to support healthcare professionals in delivering high-quality patient care.
Designed for adult, pediatric, and neonatal patients, the Cardio Care K12 and K15 offer comprehensive physiological monitoring capabilities, allowing clinicians to observe critical patient parameters in real time. Standard monitoring functions include ECG, heart rate, respiration, oxygen saturation (SpO₂), pulse rate, non-invasive blood pressure (NIBP), and temperature. Additional optional modules such as EtCO₂, IBP, cardiac output monitoring, 12-lead ECG, and cerebral state monitoring can further expand the clinical capabilities of the system.
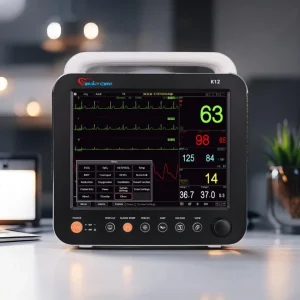
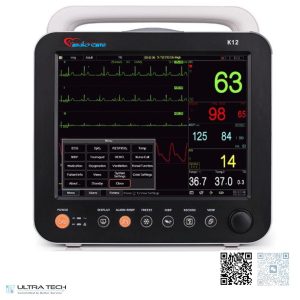
The major difference between the two models lies primarily in display size. The K12 features a high-resolution 12.1-inch display, while the K15 offers a larger 15-inch display that provides enhanced visibility of waveforms, trends, and patient information. Both systems support multiple display modes, allowing clinicians to customize screen layouts according to clinical requirements and monitoring preferences.
One of the key strengths of the Cardio Care K12 and K15 monitors is their comprehensive multi-parameter monitoring capability. The systems continuously measure and display:
- ECG and Heart Rate
- SpO₂ (Oxygen Saturation)
- Pulse Rate
- Non-Invasive Blood Pressure (NIBP)
- Respiration Rate
- Temperature
These parameters provide healthcare professionals with a complete overview of patient condition and help support rapid clinical decision-making in both routine and critical care environments.
The monitors are equipped with advanced ECG analysis features including arrhythmia detection and ST-segment analysis. Up to 28 types of arrhythmias can be detected and analyzed automatically, helping clinicians identify potentially life-threatening cardiac abnormalities at an early stage. The systems also support multiple ECG lead configurations and optional 12-lead ECG functionality for advanced cardiac assessment.
The Cardio Care K12 and K15 provide excellent waveform visualization, displaying up to 9 waveforms simultaneously and supporting expansion to as many as 13 waveforms depending on configuration. This capability enables clinicians to monitor multiple physiological signals at once without switching between screens, improving efficiency and patient safety.
Advanced NIBP technology allows automated blood pressure measurement with customizable measurement intervals. Users can configure multi-phase NIBP monitoring cycles according to clinical needs, ensuring continuous and accurate blood pressure assessment for patients requiring close observation.
The systems also feature intelligent alarm management with visual and audible alarms that alert medical staff to abnormal patient conditions. Adjustable alarm limits help healthcare providers respond quickly to critical changes in vital signs and improve overall patient safety.
For healthcare facilities seeking integration with modern hospital infrastructure, the Cardio Care K12 and K15 support advanced connectivity features including data export, software upgrades, central monitoring station connectivity, HL7 communication protocols, and bed-to-bed viewing functions. These capabilities help streamline workflow, improve patient data management, and support efficient hospital-wide monitoring systems.
The monitors incorporate ergonomic design principles with touch-screen operation options, intuitive user interfaces, and high-resolution displays that enhance ease of use. Clinical staff can quickly access patient information, review trends, configure monitoring settings, and respond to alarms efficiently, even in busy healthcare environments.
Battery backup functionality ensures continued monitoring during patient transport or temporary power interruptions. This portability makes the K12 and K15 suitable for emergency situations, intra-hospital transport, and mobile patient care applications. The systems are engineered to deliver stable performance and dependable monitoring under demanding clinical conditions.
The Cardio Care Patient Monitor K12 and K15 are ideal for:
- Intensive Care Units (ICU)
- Coronary Care Units (CCU)
- Operating Rooms (OT)
- Emergency Departments
- Recovery Rooms
- High Dependency Units (HDU)
- General Hospital Wards
- Ambulatory Care Centers
- Diagnostic and Specialty Clinics
In Bangladesh, Ultratech BD supplies the Cardio Care Patient Monitor K12 and K15 with installation support, user training, warranty coverage, preventive maintenance services, and technical assistance. This ensures healthcare facilities receive reliable monitoring solutions backed by professional after-sales support.
Overall, the Cardio Care Patient Monitor K12 and K15 provide a powerful combination of advanced monitoring technology, clinical versatility, intelligent alarm systems, comprehensive patient assessment tools, and dependable performance. Their ability to support continuous monitoring of critical physiological parameters makes them valuable assets for hospitals and healthcare facilities focused on delivering safe, efficient, and high-quality patient care.
A patient monitor is a medical device that continuously tracks and displays a patient’s vital signs (like ECG, heart rate, SpO2, blood pressure, temperature) in real-time, providing crucial data for clinicians in various settings (OR, ICU, general wards) to assess patient status, detect changes, and make informed treatment decisions, with options ranging from bedside units to portable or wireless systems.
Key Functions & Parameters of Cardio Care Patient Monitor
- ECG/EKG: Monitors heart’s electrical activity (rate, rhythm).
- SpO2(Oxygen Saturation): Measures blood oxygen levels.
- NIBP(Non-Invasive Blood Pressure): Measures blood pressure without a cuff.
- Respiration (RESP):Tracks breathing rate.
- Temperature (TEMP):Measures body temperature.
- EtCO2(Capnography): Measures carbon dioxide in exhaled breath (optional).
- IBP(Invasive Blood Pressure): Continuous pressure monitoring during surgery (optional).
Types & Applications of Cardio Care Patient Monitor
- Bedside Monitors: Standard for ICUs, ERs, and general wards.
- Central Monitors: Display multiple patients’ data from a central station.
- Transport Monitors: Portable for moving patients within the hospital.
- Wireless/Wearable Monitors: Allow patient mobility while transmitting data.
- Holter Monitors/Event Recorders: Wearable devices for long-term heart monitoring.
Importance in Healthcare
- Early Warning: Detects deterioration before it becomes critical.
- Informed Decisions: Provides real-time data for faster, better care.
- Data Management: Helps manage large amounts of patient data efficiently.
- Alarms: Alerts staff to critical vital sign change
K-10,12,15 inch Patient Monitor
10,12,15 inch high resolution touch screen with multiple display modes
Standard Parameters: 3/5 lead ECG, RESP, SpO2, PR, NIBP, TEMP
Optional Parameters: 4-IBP, EtCO2, Nellcor SpO2, SunTech NIBP, 12-lead ECG, Cardiac Output, Cerebral State Monitoring
28 types of arrhythmia and S-T segment analysis
2 year warranty on main unit and half year warranty on accessories
K-10,12,15 inch Patient Monitor
K-10,12,15 Features
• 10”, 12” 15″ high-resolution display Touch screen optional
• 9 traces on-screen waveforms and maximal up to 13
• User customized NIBP measuring cycles up to 5-phase
• Data export and software upgrade
• Versatile clinical calculations for application convenience
• HL7 protocol, Bed to bed view and 5-lead ECG available
Standard accessories–
Adult SpO2 probe
– NIBP adult cuff
– SpO2 extension cable
– ECG cable
– Skin temperature probe
– Power supply cable
– Re-chargeable lithium battery
– Dust-protective cover
– Disposable electrode
– Manual: GB, FR, IT, ES.
– PC software: GB, IT, ES
Warranty & Customer Support in Bangladesh
Ultratech provides installation, servicing, technical support and nationwide delivery in Bangladesh.
FDA Official Website
Ultratech Official Website
Frequently Asked Questions (FAQ) – Cardio Care Patient Monitor K12 & K15
Q1: What are the Cardio Care Patient Monitor K12 and K15 ?
A: The Cardio Care K12 and K15 are advanced multi-parameter patient monitoring systems designed to continuously monitor vital signs such as ECG, heart rate, SpO₂, NIBP, respiration, temperature, and other optional parameters in hospitals and critical care environments.
Q2: What is the difference between the Cardio Care Patient Monitor K12 and K15 models?
A: The primary difference is the display size. The K12 features a 12.1-inch display, while the K15 comes with a larger 15-inch display for enhanced waveform visibility and patient data monitoring. Both models offer similar monitoring capabilities and clinical functions.
Q3: Which parameters can be monitored using the Cardio Care Patient Monitor K12 and K15?
A: Standard monitoring parameters include:
- ECG
- Heart Rate (HR)
- SpO₂ (Oxygen Saturation)
- Pulse Rate (PR)
- Non-Invasive Blood Pressure (NIBP)
- Respiration Rate (RESP)
- Temperature (TEMP)
Optional parameters may include:
- EtCO₂
- Invasive Blood Pressure (IBP)
- Cardiac Output (CO)
- 12-Lead ECG
Q4: Are the monitors suitable for adult, pediatric, and neonatal patients?
A: Yes. The Cardio Care K12 and K15 support monitoring for adult, pediatric, and neonatal patients, making them versatile solutions for various clinical departments.
Q5: Can the Cardio Care Patient Monitor be used in Intensive Care Units (ICU)?
A: Yes. The K12 and K15 are specifically designed for ICU, CCU, emergency departments, operating rooms, and other critical care environments where continuous patient monitoring is essential.
Q6: Do these monitors provide ECG monitoring?
A: Yes. Both models offer real-time ECG monitoring with advanced waveform display and cardiac rhythm analysis to support comprehensive cardiac assessment.
Q7: What is arrhythmia analysis?
A: Arrhythmia analysis automatically detects abnormal heart rhythms and alerts healthcare professionals to potential cardiac abnormalities, allowing faster clinical intervention when necessary.
Q8: Can the Cardio Care Patient Monitor K12 and K15 measure blood oxygen saturation (SpO₂)?
A: Yes. The monitors provide continuous SpO₂ monitoring, allowing clinicians to evaluate oxygen levels and respiratory function accurately.
Q9: Do the monitors support non-invasive blood pressure measurement?
A: Yes. Both models include automatic NIBP monitoring with configurable measurement intervals and trend analysis.
Q10: Is temperature monitoring available?
A: Yes. Temperature monitoring is included, allowing healthcare providers to continuously assess patient body temperature during treatment and recovery.
Q11: Can the monitors display multiple waveforms simultaneously?
A: Yes. The systems can display multiple waveforms and numerical parameters simultaneously, providing comprehensive patient monitoring on a single screen.
Q12: Do the monitors include alarm functions?
A: Yes. The K12 and K15 feature visual and audible alarms that notify healthcare staff when patient parameters exceed predefined safety limits.
Q13: Can alarm limits be customized?
A: Yes. Healthcare professionals can configure alarm settings according to patient condition and clinical requirements.
Q14: Is battery backup available?
A: Yes. The monitors include an internal rechargeable battery that supports continuous monitoring during patient transport and temporary power interruptions.
Q15: Can the monitors be used during patient transportation?
A: Yes. Their portable design and battery operation make them suitable for intra-hospital patient transport and emergency situations.
Q16: Do the monitors support central monitoring systems?
A: Yes. The K12 and K15 can connect to central monitoring stations, allowing clinicians to monitor multiple patients from a centralized location.
Q17: Are the monitors compatible with hospital information systems?
A: Many configurations support connectivity with hospital networks, central monitoring systems, and data management platforms to improve workflow efficiency.
Q18: What clinical departments commonly use the Cardio Care Patient Monitor K12 and K15?
A: These monitors are commonly used in:
- Intensive Care Units (ICU)
- Coronary Care Units (CCU)
- Operating Theaters (OT)
- Emergency Rooms (ER)
- Recovery Rooms
- High Dependency Units (HDU)
- General Wards
- Specialty Clinics
Q19: Are the monitors easy to operate?
A: Yes. The user-friendly interface, intuitive controls, and high-resolution display allow healthcare providers to operate the system efficiently with minimal training.
Q20: Can patient data and trends be reviewed?
A: Yes. The monitors store patient data, trend graphs, and event records, allowing clinicians to review patient history and monitor treatment progress.
Q21: How accurate are the Cardio Care Patient Monitors 12 and K15 ?
A: The monitors utilize advanced monitoring technologies and signal processing algorithms to deliver accurate and reliable measurements across multiple parameters.
Q22: Do the monitors support future upgrades?
A: Depending on configuration, additional modules and software upgrades may be available to expand monitoring capabilities and meet evolving clinical needs.
Q23: Does Ultratech BD provide installation and training?
A: Yes. Ultratech BD offers professional installation, user training, application support, and guidance to ensure optimal monitor performance.
Q24: Is technical support available in Bangladesh?
A: Yes. Ultratech BD provides preventive maintenance, troubleshooting, spare parts support, and technical service throughout Bangladesh.
Q25: Why should healthcare facilities choose the Cardio Care Patient Monitor K12 and K15 ?
A: The Cardio Care K12 and K15 offer comprehensive vital sign monitoring, advanced alarm management, reliable performance, user-friendly operation, and excellent value for hospitals and healthcare providers. Their flexibility, accuracy, and clinical versatility make them ideal solutions for modern patient monitoring requirements.
FDA
The Food and Drug Administration (FDA) is a federal agency of the United States Department of Health and Human Services (HHS). The FDA is responsible for protecting and promoting public health through the control and supervision of food safety, tobacco products, caffeine products, dietary supplements, prescription and over-the-counter pharmaceutical drugs (medications), vaccines, biopharmaceuticals, blood transfusions, medical devices, electromagnetic radiation emitting devices (ERED), cosmetics, animal foods & feed, and veterinary products.
The FDA’s primary focus is enforcing the Federal Food, Drug, and Cosmetic Act (FD&C). However, the agency also enforces other laws, notably Section 361 of the Public Health Service Act as well as associated regulations. Much of this regulatory-enforcement work is not directly related to food or drugs but involves other regulated products like lasers, cellular phones, and condoms. In addition, the FDA uses its authority to mitigate diseases in a variety of contexts, from household pets to human sperm donated for use in assisted reproduction.
The FDA is led by the commissioner of food and drugs, who is appointed by the president with the advice and consent of the Senate. The commissioner reports to the secretary of health and human services. Marty Makary is the current commissioner.
The FDA’s headquarters is located in the White Oak area of Silver Spring, Maryland, occupying the former Naval Ordnance Laboratory The agency has 223 field offices and 13 laboratories located across the 50 states, the United States Virgin Islands, and Puerto Rico. In 2008, the FDA began to post employees to foreign countries, including China, India, Costa Rica, Chile, Belgium, and the United Kingdom.










Reviews
There are no reviews yet.